|
11/19/2022 0 Comments Half life chemistry calculator
Enter the values below to calculate Half-Life. Half life chemistry calculator how to#how to evaluate natural-logarithm powers on your calculator (for many calculators. Probabilistic nature A half-life usually describes the decay of discrete entities, such as radioactive atoms.įind the Half-Life of any number for free. An interesting and useful aspect of radioactive decay is half-life. The accompanying table shows the reduction of a quantity as a function of the number of half-lives elapsed. Click the Calculate button, and the calculator will fill in the missing. 
Half-life is constant over the lifetime of an exponentially decaying quantity, and it is a characteristic unit for the exponential decay equation. Fill in three values and leave one blank. Rutherford applied the principle of a radioactive element's half-life to studies of age determination of rocks by measuring the decay period of radium to lead-206. The original term, half-life period, dating to Ernest Rutherford's discovery of the principle in 1907, was shortened to half-life in the early 1950s. This medicine half life calculator estimates the action of any medicine and the way concentration decreases in percentage in plasma according to half life and dosage. The converse of half-life is doubling time. The students have to figure out how much of a substance will be left after a given amount of time, how many half-lives have passed if there is a given quantity of the substance present, and how long it will take for a substance to decay to a given amount. For example, the medical sciences refer to the biological half-life of drugs and other chemicals in the human body. The term is also used more generally to characterize any type of exponential or non-exponential decay. Half life chemistry calculator download#You may download the Specific Activity Adjustment Calculator (Microsoft Excel. The half-life is independent of the physical state (solid, liquid, gas), temperature, pressure, the chemical compound in which the nucleus finds itself. t('1/2') - the half-life of the decaying quantity. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable atoms survive. Half life - the time it takes for a product to decay sufficiently to. 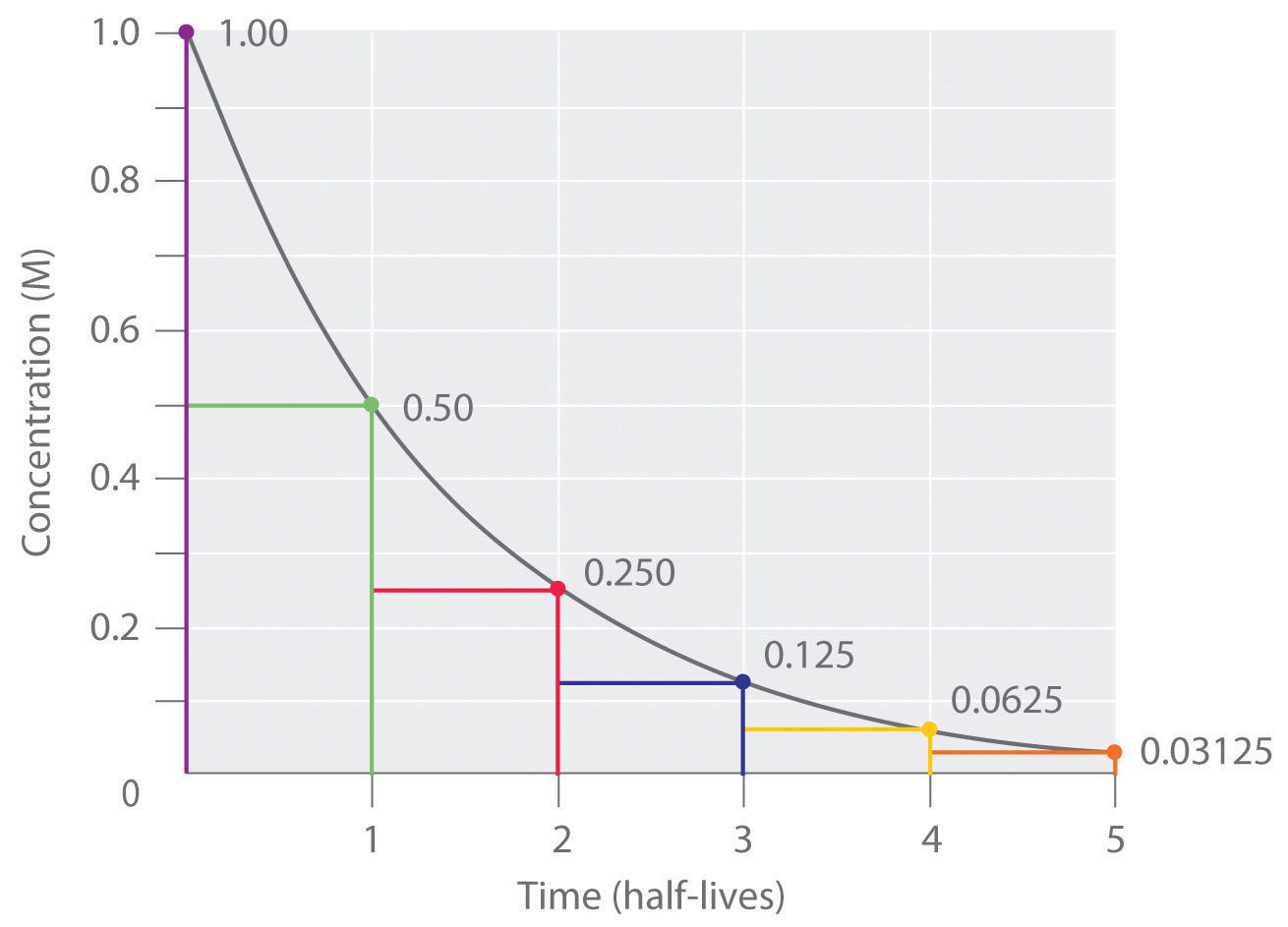 
Half-life (symbol t12) is the time required for a quantity to reduce to half of its initial value. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
